Marie Skłodowska-Curie Action ITN NANOMED
H2020 MSCA ITN
Nanomedicine: an integrative approach (NANOMED) is a Marie Sklodowska-Curie Innovative Training Network (ITN), coordinated by Prof. Jan van Hest (Radboud University, Nijmegen, The Netherlands). The project consortium is composed of 7 beneficiaries and 5 partner organizations. NANOMED will train a new generation of multi-disciplinary nanotechnology experts capable of supporting and managing the effective translation of molecular innovations into clinically applicable therapeutic solutions. Within the NANOMED framework, Early Stage Researchers (ESRs) will develop a broad understanding of the entire process of nanomedicine therapy development, from bench to bedside, including formulation, production, pharmacokinetics, toxicological and regulatory aspects. These scientists will develop unique cross-disciplinary skills in chemistry, pharmacology and chemical engineering at GMP level to allow them to develop the effective, safe and efficiently producible nanomedicines of the future.
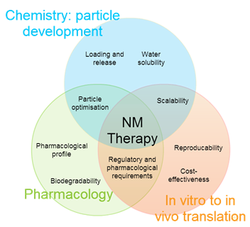
Reasons that explain the current moderate success of NM are: (1) promising preclinical results are often poorly predictive for clinical safety and effectiveness, (2) the efficient, scalable and reproducible GMP production of nanocarriers has proven to be challenging and (3) regulatory frameworks are not yet fully equipped to efficiently facilitate the introduction of novel nanomedicines.
These obstacles are often encountered since the developmental process from carrier design to clinical assessment is performed by a range of scientists from different backgrounds who have difficulty interacting and communicating with each other to clearly understand the necessary design criteria and the scope and limitations of NM.
NANOMED brings together all the necessary expertise to oversee the entire development trajectory required for NM. This is achieved by the combined effort of 7 beneficiaries from academia and industry and 5 non-academic partner organisations, which are all thoroughly rooted in nanosciences and pharmaceutical sciences. Our general objective is to develop scalable and highly controllable design and synthesis methods for the most promising nanomedicine types in a preclinical setting. NANOMED will train the next generation of NM scientists by offering an extensive joint training programme to 15 incoming ESRs. It focuses on promoting scientific excellence and exploits the specific research and commercial expertise and infrastructure of the NANOMED network as a whole. The exposure to all elements of NM design enables NANOMED to translate expertise from all disciplines to the ESRs, to educate the future leading scientists in the NM field.
Objectives
These are the scientific objectives of NANOMED:
- To develop scalable routes for the production of biodegradable nanocarriers
- To formulate the carriers with appropriate drugs and study drug release, biodegradation and toxicology
- To investigate the particle fate upon cell uptake with advanced imaging methodologies
- To pursue particle functionalisation in order to attain targeted delivery of drugs.
- To apply both mathematical and animal models to study the distribution and release kinetics of the nanocarrier systems
- To investigate the nanocarrier applicability with regard to sterilisation potential, shelf life, regulatory and safety issues and clinical trials
Role of Ghent University
- Supervision of 3 ESR candidates in their PhD research
- Organization of a basic training course (Pharmacology for chemists)
- Organization of an advanced training workshop (Intraperitoneal drug delivery) covering the methodologies available to assess the efficacy of intraperitoneal drug delivery approaches.
Contact
Prof. Stefaan De Smedt
Department of Pharmaceutics
Phone number: + 32 (0)9 264 80 76
E-mail: stefaan.desmedt@ugent.be
Prof. Katrien Remaut
Department of Pharmaceutics
Phone number: + 32 (0)9 264 80 46
E-mail: katrien.remaut@ugent.be
Dr. Koen Raemdonck
Department of Pharmaceutics
Phone number: + 32 (0)9 264 80 78
E-mail: koen.raemdonck@ugent.be