Cardiac and Arterial shear wave elastography
Cardiac shear wave elastography
- Achieve mechanical insights in in vivo cardiac SWE
- Assess the performance and sensitivity of SWE
- Improve current shear wave speed estimation techniques or propose new or more advanced material characterization methods for SWE
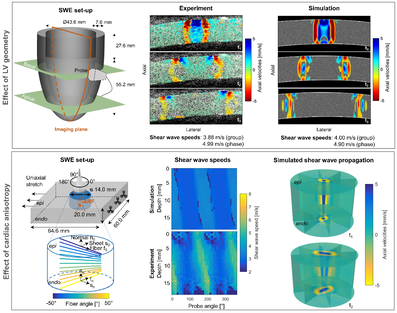
Figure 1: Modeling ARF-based shear wave propagation in simplified cardiac settings.
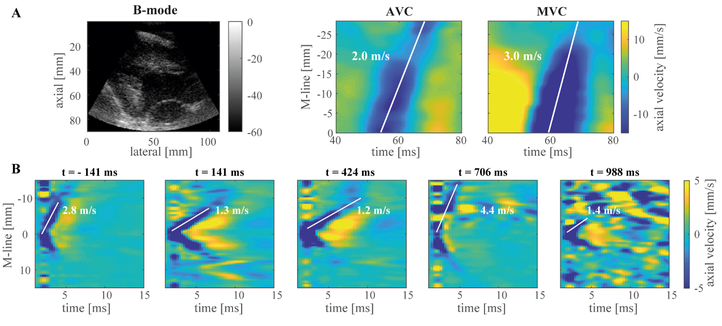
Next to ARF-based SWE, we are also currently working on better understanding cardiac natural SWE. Natural SWE studies the mechanical waves traveling along the interventricular septum after valve closure. The major advantage of these natural shear waves compared to ARF-induced waves is that no high-intensity ultrasound beam is needed (leading to acoustically safer acquisitions) and that the signal-to-noise ratio of these acquisitions is higher than that of ARF-based SWE (improving the accuracy and robustness post-processing). However, the properties of the excitation source cannot be controlled in natural SWE. In collaboration with the Thorax center BME (Erasmus MC, Rotterdam, Netherlands) and the Cardiovascular Imaging and Dynamics lab (KULeuven, Leuven, Belgium), we are currently investigating the link between natural and ARF-based SWE (see Fig. 2).
Figure 2: Natural vs. acoustic radiation force based shear wave elastography. Panel A depicts shear wave propagation after aortic and mitral valve closure (AVC and MVC respectively). Panel B shown shear wave propagation for ARF-based SWE at different time points throughout the cardiac cycle. Adapted from.
Arterial shear wave elastography
Having prior knowledge about changes in arterial wall material properties (stiffening of the wall) in patients could help doctors predict such a commonly wide-spread disease like atherosclerosis and, as a consequence, prevent other health- and life-threatening conditions: brain stroke or myocardial infarction. Nowadays, there is still lack of a technique in common clinical practice, that could estimate arterial wall stiffness directly and accurately, and being able to detect small, early-stage disease development.
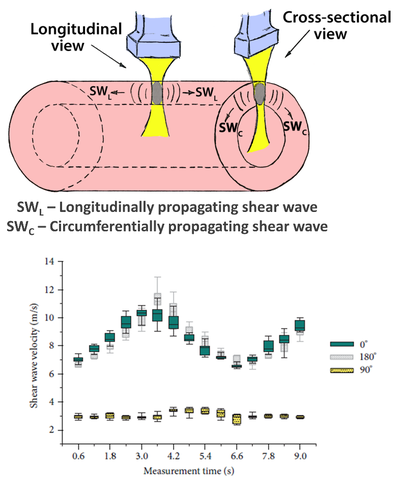
Supersonic shear wave imaging technique in arterial applications could fill this niche. The principal concept behind the technique is straightforward and smart: you apply a small force onto the arterial wall by focused ultrasound. This force, called acoustic radiation force, creates secondary waves, called shear waves, which propagate with a certain speed. By measuring their propagation speed, we can deduce the material properties of the arterial wall (i.e. stiffness).
Unfortunately, in practice it is more complicated than that: due to the small thickness of arterial walls, such induced shear waves behave as guided waves, making the conversion between their speed and arterial elasticity complex. To be able to correctly predict arterial stiffness by using this technique, we have to improve our understanding of the nature of these waves in arterial wall settings.
The PhD of Darya Shcherbakova focuses on series of controlled experiments on phantom materials and ex-vivo equine aortic tissues using both supersonic shear wave imaging technique and a mechanical testing machine to measure tissue stiffness (see Fig. 3). Afterwards, we tried to model the same experiment numerically and gain more insight into the propagation of the resulted shear waves and the dependence of the shear wave speed on the arterial geometry and intrinsic material properties: anisotropy and non-linearity.
You can find more about these experiments and general background information in the following articles [11-17].
References
-
A. Caenen, et al., A versatile and experimentally validated finite element model to assess the accuracy of shear wave elastography in a bounded viscoelastic medium, IEEEE TUFFC, 2015.
-
A. Caenen, et al., Myocardial stiffness assessment in pediatric cardiology using shear wave imaging, IEEE IUS, 2015.
-
A. Caenen, et al., Finite element simulations to support the measurement and analysis of shear wave dispersion, IEEE IUS 2016.
-
A. Caenen, et al., Investigating shear wave physics in a generic pediatric left ventricular model via in vitro experiments and finite element simulations, IEEE TUFFC, 2017.
-
A. Caenen, et al., Effect of ultrafast imaging on shear wave visualization and characterization: an experimental and computational stud in a pediatric ventricular model, Applied Sciences, 2017.
-
A. Caenen, et al., The effect of stretching on transmural shear wave anisotropy in cardiac shear wave elastography, IEEE IUS, 2017.
-
A. Caenen, et al., An in silico framework to analyze the anisotropic shear wave mechanics in cardiac shear wave elastography, PMB, 2018.
-
A. Caenen, et al., Investigating the degree of shear wave speed anisotropy as a function of studied ventricular zone, IEEE IUS, 2018.
-
A. Caenen, et al., Analyzing the shear wave mechanics in cardiac shear wave elastography using finite element simulations, IEEE IUS, 2018.
-
Keijzer, et al., A comparison of natural and acoustic radiation force induced shear wave propagation speed measurements in open-chest pigs, IEEE IUS, 2019.
-
D. Shcherbakova, et al., Supersonic shear wave imaging to assess arterial anisotropy: ex-vivo testing of the horse aorta, IEEE IUS, 2013.
-
D. Shcherbakova, et al., Supersonic shear wave imaging to assess arterial nonlinear behavior and anisotropy: proof of principle via ex vivo testing of the horse aorta, Advances In Mechanical Engineering, 2014.
-
D. Shcherbakova, et al., Experimental study on the effect of the cylindrical vessel geometry on arterial shear wave elastography, IEEE IUS, 2015.
-
D. Shcherbakova, et al., A finite element model to study the effect of tissue anisotropy on ex vivo arterial shear wave elastography measurements, PMB, 2017.
-
A.P. Sarvazyan, et al., Biophysical bases of elasticity imaging, Acoustical Imaging, 1995.
-
A.P. Sarvazyan, et al., Shear wave elasticity imaging: a new ultrasonic technology of medical diagnostics, UMB, 1998.
-
M. Couade, et al., Quantitative assessment of arterial wall biomechanical properties using shear wave imaging, UMB, 2010.
IBiTech researchers currently active on the project
Funding sources
Junior post-doctoral fellowship by Research Foundation Flanders FWO – grant 1211620N 2019-2022 (Annette Caenen)
PhD fellowship by Flanders Innovation and Entrepreneurship Agency VLAIO – grant 141010 2014-2019 (Annette Caenen)
Post-doctoral fellowship by Research Foundation Flanders FWO – grant 1276416N 2014-2016 (Abigail Swillens)
PhD fellowship by Research Foundation Flanders FWO – grant 11U2116N 2013-2018 (Darya Shcherbakova)
Finalized PhDs within IBiTech
A Biomechanical Analysis of Shear Wave Elastography in Pediatric Heart Models (Annette Caenen)
Relevant link