Marie Skłodowska-Curie Action IF ImmunoFarm
H2020 MSCA IF
IgA-based oral therapy for protection of piglets against infection with enterotoxigenic E. coli
Duration in months 24
Coordinator: VIB
Listof Participants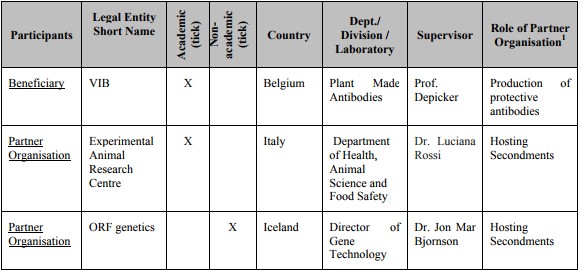
It has been since the 70s that the world’s pork industry has been on rise, and global production has already surpassed 110 million tons per year. Pork is the world’s principal meat product by a significant margin surpassing both chicken and beef. Post-weaning diarrhoea (PWD) caused by enterotoxigenic Escherichia coli (ETEC) carrying the F4 (ETEC-F4) or F18 (ETEC-F18) fimbriae, is one of the most important global issues leading to huge economic losses in the worldwide porcine rearing industry. This repeatedly occurring disease affects piglets generally the first week after the weaning phase. Economic losses are generated due to a decrease in the weight gain and, most importantly, a mortality of ̴ 2% - 7% piglets/year (annual loss of about €15.000/500-sow herd). Antibiotics have been generally used as prophylactic solutions, however, their use have been recently banned in Europe due to an increasing occurrence of antibiotic resistant E. coli strains. However the therapeutic use of antibiotics against PWD is prevalent and should be reduced, as stated by the European Commission. It is well-known that the prevention of enteric infections relies on the mucosal immunity. Yet, the development of oral vaccines is hindered by the neutralization of the vaccine with pre-existing maternal antibodies in the consumed milk. Moreover, vaccines administered after weaning do not provide an immediate protection, as they require time to induce immunity at the mucosal surface. Therefore, there is a gap of two or three weeks in which piglets would not be protected against PWD.
A promising complement or even alternative to antibiotics and vaccines for prophylaxis and treatment of enteric diseases is the oral delivery of recombinant antibodies. Passive immunisation (PI) can be used when immediate immunity is needed.
The best suited antibody isotype for passive immunotherapy is IgA, the most abundant isotype in the mucosal immune system. IgA exists naturally in it secretory form (SIgA) in the mucosal surfaces, however, it has been shown in our lab (Virdi et al. in preparation) that the use of monomeric IgA is as efficient as the use of sIgA for oral delivery against ETEC infections. Therefore, the design and production of the antibodies against ETEC-F18 in this project has been focused on simplified monomeric IgA versions. Plant-expression systems are an attractive platform for production of IgA antibody cocktails. They are cost-effective, highly scalable and have a low risk of contamination with mammalian pathogens.
The ideal platform for production of antibodies aimed at oral passive immunization for veterinary, and even medical uses is such that can produce bulk quantities in short time, does not need expensive purification or formulation and can be stock-piled without the need of refrigeration. Thus, commercial seed crops are ideal cost-effective production platforms and offer the added benefits of being a natural source of protein and providing a stable environment for storage and maintenance. Moreover, this proof of concept might as well be very valuable for public health emergencies such as viral outbreaks, because large quantities of antibodies could be stock-piled in a ready-to-use formulation and could be quickly delivered to the population if required, without the need of syringes or medical instrumental, just as a food additive. Soybean being a commercial crop can be scaled up at a cost effective scale and is an important source of protein used in animal feed, demonstrating its convenience to be used as a feed additive.
With our ImmunoFarm project, and together with a partner project in the laboratory, not only we have generated a feed complement that is currently being evaluated by a company for its future commercialization, but also, we have paved the way to a great variety of uses of in-seed production of antibodies aimed for oral passive immunization, which will be of great socio-economic impact.
During the course of the project the strategies followed changed slightly from the ones described in the project proposal. In summary, four different anti ETEC-F18 antibodies in a simplified IgA-like format were engineered from phage display-selected variable domains (VHHs). The synergistic vs. competitive effect of different antibody versions was evaluated by means of a competitive ELISA setting. The different antibodies were first produced in the seeds of Arabidopsis thaliana transformants. An in vivo ETEC-F18 challenge was then performed using four groups of piglets, two of them receiving a cocktail of two elite antibodies in two different concentrations, one receiving only one of the elite antibodies and a control group receiving regular feed (negative control group). Last, the IgA antibodies were also transferred to soya seeds as production platform.
Objectives
These are the objectives of ImmunoFarm:
The main objective was to generate a cost-effective prophylactic product intended to prevent PWD in piglets. The objective has been accomplished. The product that we have generated consists of four light chain-devoid VHH-IgA antibodies directed against ETEC-F18. For up-scaling, each antibody has been produced in the seeds of transgenic soya. Thus, the product can then be formulated to include crushed seeds of each antibody producing line in quantified and reproducible proportions.
Further in vivo tests need to be performed in order to bring such product to the market. Also a better understanding of the cleavage profile needs to be achieved prior to commercialization.
Role of Ghent University
The role of VIB was to Coordinate the project. The project was carried out in the facilities of VIB-UGent.
Contact
Prof. Ann Depicker
Department Plant Systems Biology
Phone number: +32 9 331 39 40
E-mail: anpic@psb.vib-ugent.be